Atomo Diagnostics HIV test to be more widely available
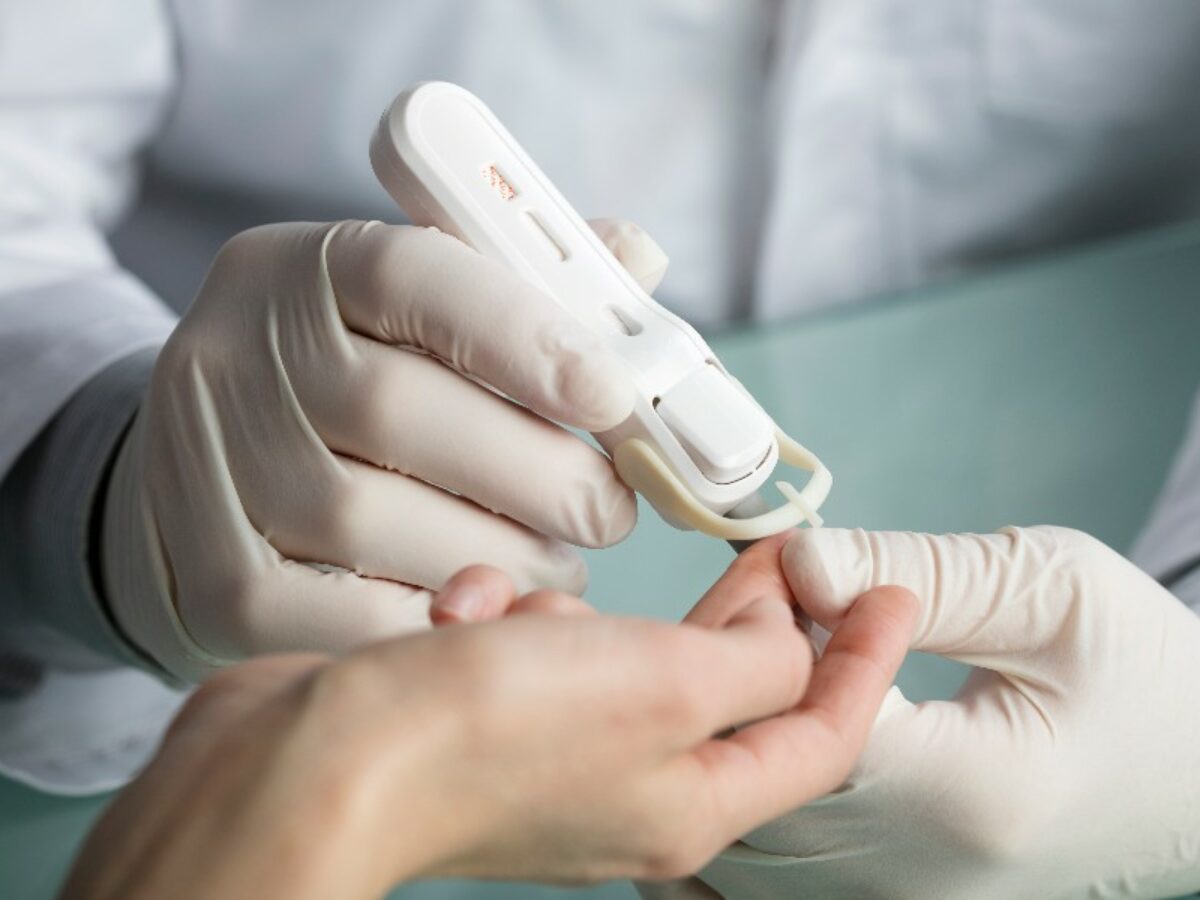
Rapid HIV test manufacturer Atomo Diagnostics will have its handheld test device more widely available in low and middle-income countries (LMICs) through an agreement with healthcare company Viatris Inc.
Sydney's Atomo has agreed to a multi-year expoansion of HIV tself-testing using its kits through global health agency Unitad and Viatris'subsidiary Mylan.
The Mylan HIV Self-Test will be distributed in 135 countries by Viatris.
Atomo Diagnostics' test, manufactured in Australia, detects the presence or absence of HIV antibodies in a small blood sample obtained from a fingertip.
Pre-qualified by the World Health Organisation, the test is an integrated handheld device that reduces common sources of user error.
Atomo managing director John Kelly said the agreement would bring a material step-up in demand for HIV self-tests globally.
Kelly said: “This demand enables Atomo to achieve sustainable operational and supply chain efficiencies, allowing us to support Viatris in its discussions with Unitaid.
“(This will) significantly expand the rollout of HIV self-testing in LMICs in the coming years.”
Unitaid is a global health agency engaged in finding innovative solutions to prevent, diagnose and treat diseases more quickly, cheaply and effectively, in low-and middle-income countries.
Viatris' Rakesh Bamzai said the agreement would contribute to the United Nations goal to educate 90 per cent of people of their HIV status.
Bamzai said: “We have been on the frontlines of the global fight against HIV/AIDS for more than a decade and understand the importance of both prevention and treatment.”
Picture: MTP Connect/Atomo Diagnostics
Subscribe to our free @AuManufacturing newsletter here.
Topics Technology
@aumanufacturing Sections
Analysis and Commentary Awards casino reviews Defence Gambling Manufacturing News Online Casino Podcast Technology Videos

