Breaking into new grounds: Orthocell eyes $75M Canadian market with game-changing nerve repair product
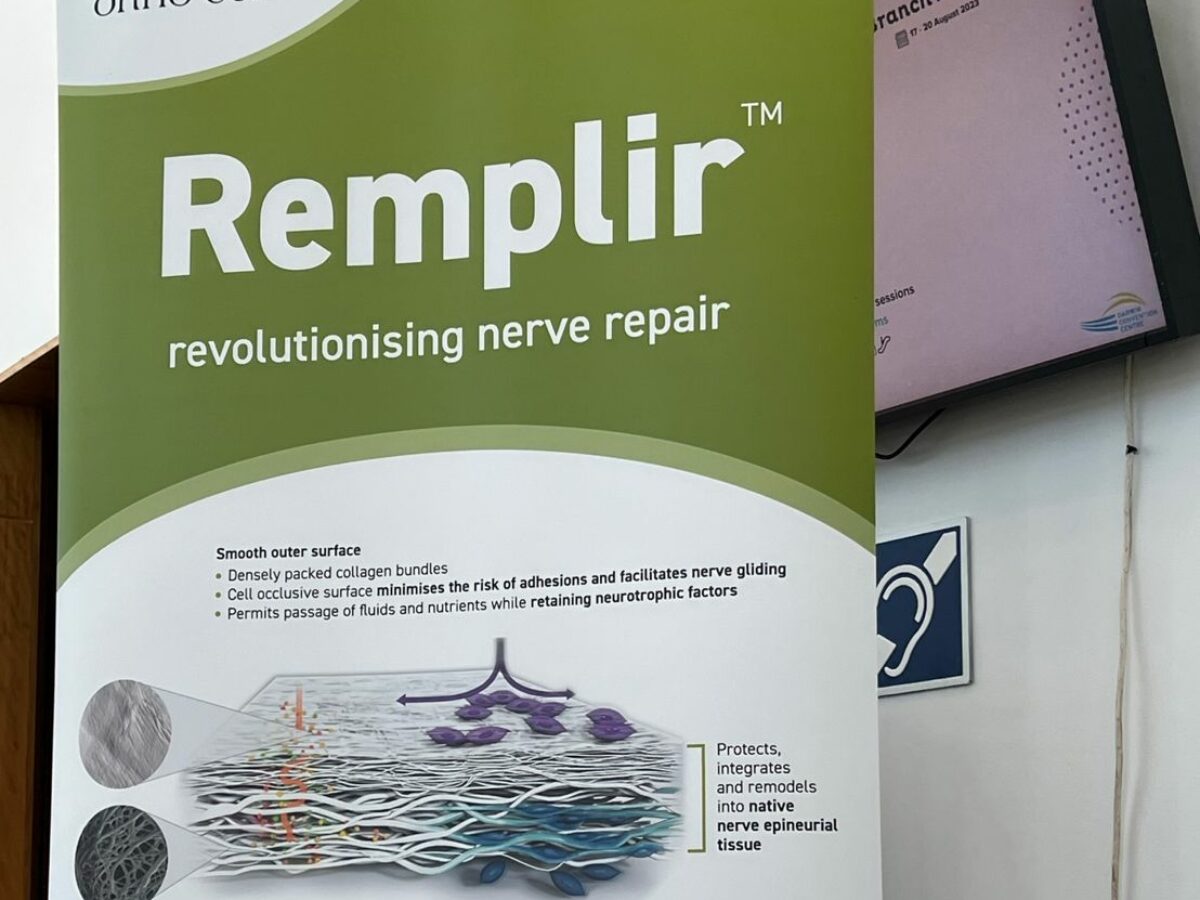
In a bold move to capture a significant share of the global nerve repair market, Orthocell, a pioneer in regenerative medicine, has submitted a regulatory application to Health Canada for its leading product, Remplir. This step marks a strategic expansion into the Canadian market, valued at approximately US$75 million, with approvals anticipated in the latter half of 2025.
Remplir, a collagen wrap designed for nerve repair surgeries, has demonstrated impressive sales traction and consistent revenue growth since its launch in November 2022. It is currently utilised by around 180 orthopedic and plastic surgeons across Australia, New Zealand and Singapore.
The Canadian approval is just the beginning of Orthocell's global rollout planned for 2025, which includes entering markets in Thailand, the United Kingdom, European Union and Brazil. With the U.S. market clearance expected by early April 2025, Orthocell is strategically positioning itself to tap into the larger US$1.6 billion U.S. nerve repair market.
Orthocell's proactive international expansion strategy is underpinned by a robust financial standing, with approximately AU$33 million in cash reserves, ensuring the company is well-equipped to scale operations and increase its market presence globally. With an eye on capturing a 20 per cent market share of the estimated US$3.2 billion available in targeted markets, Orthocell is set to significantly boost its revenue and market influence.
As the company awaits regulatory nods, preparations for the U.S. market launch are already underway, further demonstrating Orthocell's commitment to bringing innovative medical solutions to a global audience. This strategic expansion not only enhances Orthocell's commercial footprint but also promises substantial growth potential in the burgeoning field of nerve repair.
Pic sourced from LinkedIn.
Further Reading
Manufacturing news briefs — stories you might have missed
Orthocell receives first US product approval
Topics Manufacturing News
@aumanufacturing Sections
Analysis and Commentary Awards casino reviews Defence Gambling Manufacturing News Online Casino Podcast Technology Videos





