Cortical Dynamics may sell in South Korea
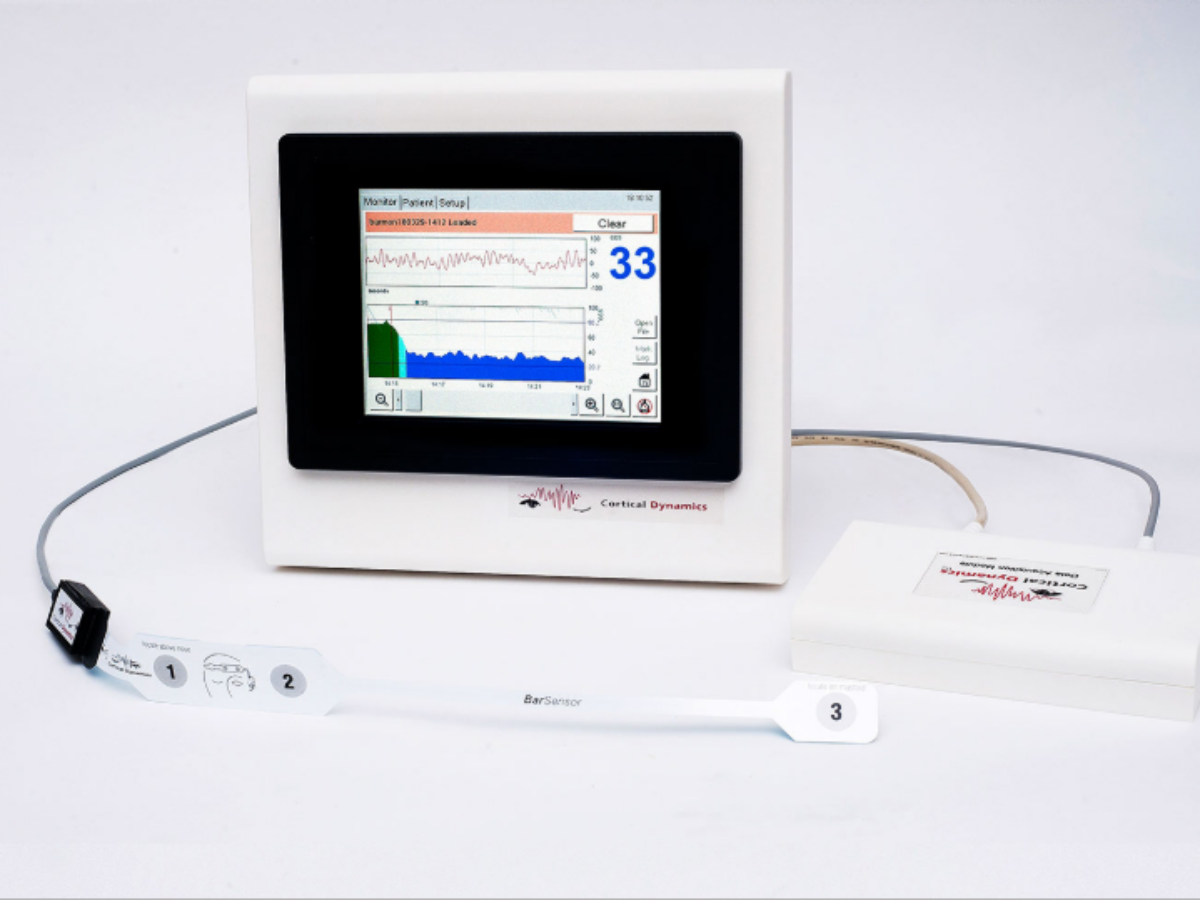
Cortical Dynamics has received regulatory approval which will lead on to an ability to sell its brain anaesthetic response monitoring (BARM) system device in South Korea.
The Perth company's Korean distributor, Globaluck, received a certificate of approval from the Korea Good Manufacturing Practice, according to an announcement by Cortical shareholder, bphenergy (ASX: BPH).
The approval is required by the country's minister of food and drug safety, and follows a visit to the company's manufacturing facility.
BARM improves on electroencephalogram (EEG) technologies by incorporating the latest advances in the understanding of how the brain’s rhythmic electrical activity (EEG) is produced.
It monitors a patient's hypnotic and analgesic state, leading to improved anaesthetic and surgical outcomes, reducing recovery times and minimising drug costs.
Cortical was founded in 2004 to commercialise technology developed at Swinburne University of Technology, Melbourne.
Picture: Cortical Dynamics
Subscribe to our fee @AuManufacturing newsletter here.
Topics Manufacturing News
@aumanufacturing Sections
Analysis and Commentary Awards casino reviews Defence Gambling Manufacturing News Online Casino Podcast Technology Videos





