Snakebite treatment stems severe bleeding
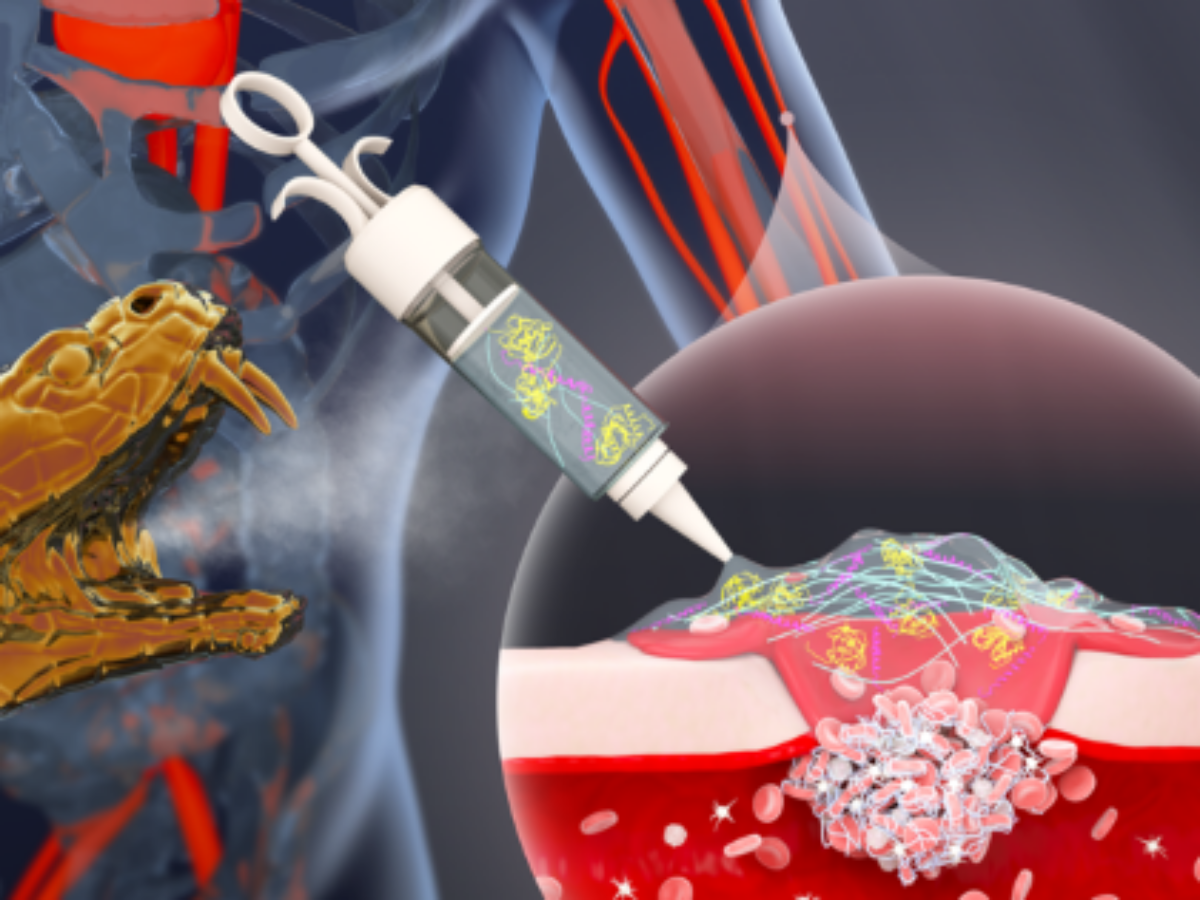
University of Queensland researchers have inspiration from nature to develop unique hydrogels derived from snakes that can save people from blood loss incurred during traumatic injury.
Based at the Australian Institute for Bioengineering and Nanotechnology (AIBN), postdoctoral research fellow Dr Amanda Kijas and Professor Alan Rowan looked at compounds in snake venom that activate the blood clotting system and block blood vessels, which can induce a stroke or heart attack.
Other better known compounds destroy capillary vessels and cause internal bleeding.
Bleeding out from traumatic injury for civilians and the military is a leading cause of preventable death worldwide, second only to brain and spinal cord injuries.
A first-aid kit offers little actual aid to a person in an emergency bleeding situation – place gauze on the wound and apply pressure, or find a tourniquet.
With support from the Australian National Fabrication Facility's Queensland Node (ANFF-Q), the team developed a potentially revolutionary wound treatment system that uses the snake venom protein, ecarin.
According to the researchers: “This begins a series of biochemical reactions that leads to the formation of fibrin, long fibres that interact with platelets to form a clot over a wound site.
“Overall, ecarin supercharges the clotting process and stems the flow of blood.”
However, triggering clot formation within a wound is limited in effectiveness if the blood clot subsequently breaks down, a process called fibrinolysis.
The researchers employed a second snake venom protein called textilinin that effectively counters fibrinolysis – a unique capability not seen in other point-of-injury wound treatment products.
The researchers developed a product in which the proteins are delivered within a topically applied, thermo-responsive hydrogel to seal the wound.
Proof-of-concept in vivo studies have demonstrated the effectiveness of their wound treatment system, achieving a striking five-fold reduction in blood loss and a three-fold reduction in clotting time.
The product has been awarded funding from the US Department of Defense, to undertake preclinical evaluations to gather more data on the safety and efficacy of this agent.
Image: ANFF
Topics Technology
@aumanufacturing Sections
Analysis and Commentary Awards casino reviews Defence Gambling Manufacturing News Online Casino Podcast Technology Videos