First human studies for Imagion cancer detection system
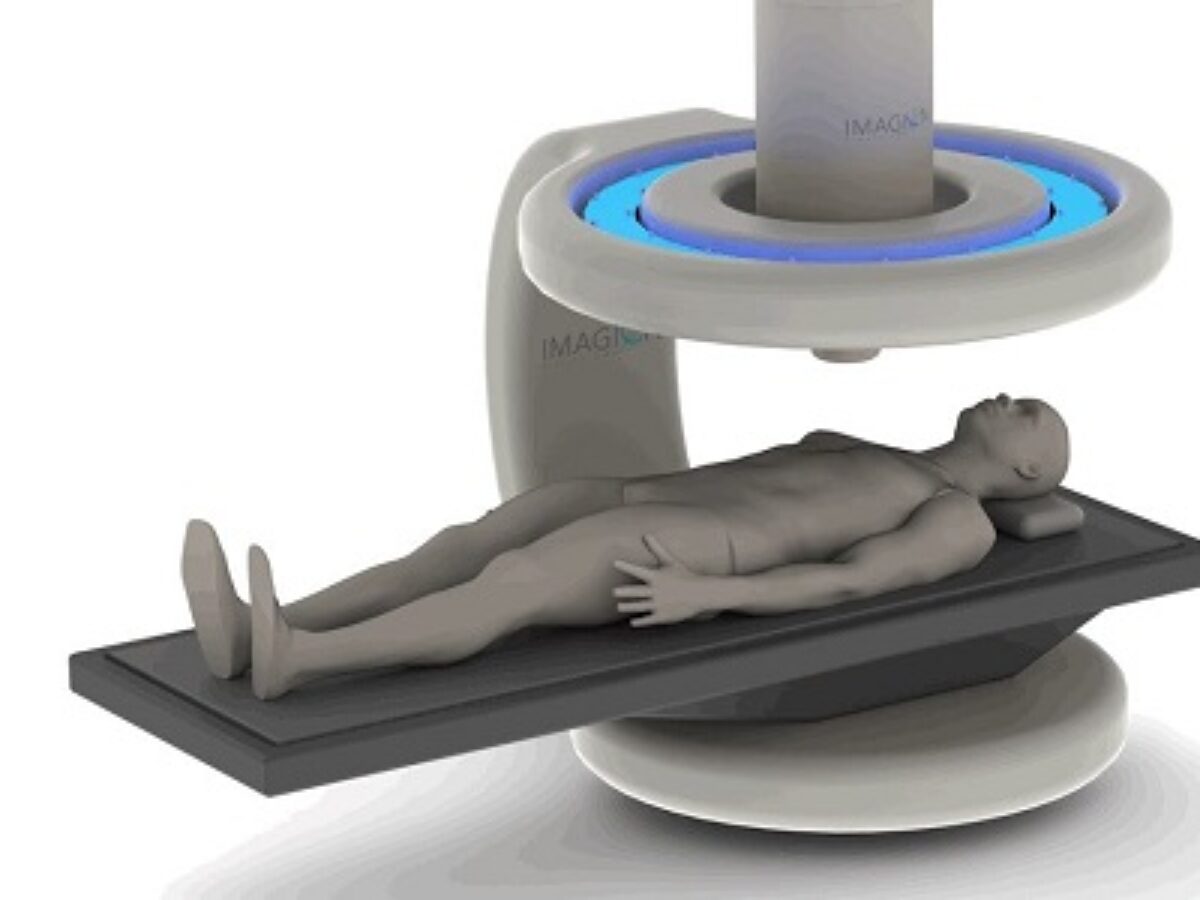
Imagion Biosystems have announced they will commence the first human trials of their MagSense cancer imaging system in Australia in the fourth quarter of 2020.
The company is focused on identifying multiple trial sites for an initial enrolment of 15 to 20 patients subject to Human Research Ethics Committee approvals.
Imagion’s MagSense is a new non-radioactive and safe diagnostic imaging technology that combines biotechnology and nanotechnology to detect cancer and other diseases.
Specialised MagSense nanoparticles are coated with tumour targeting antibodies and can be administered by simple intravenous injection.
Weak, but highly sensitive magnetic fields are used to locate the nanoparticles which bind to the targeted tumour cells.
Executive chairman Bob Proulx said the primary objective of the trials would be to demonstrate that the company's nanoparticles were safe.
He said: “It will also generate valuable data to show the effectiveness of our targeted nanoparticles in detecting the presence and spread of HER2 breast cancer using two different imaging modalities.”
In May @AuManufacturing reported that the first phase of manufacture of a nanoparticle formulation had been completed on the way to human trials.
Picture: Imagion Biosystems
Subscribe to our free @AuManufacturing newsletter here.
Topics Manufacturing News Technology
@aumanufacturing Sections
Analysis and Commentary Awards casino reviews Defence Gambling Manufacturing News Online Casino Podcast Technology Videos





